COVID-19
RAPID ANTIGEN TEST
The TESTSEALLABS™ COVID-19 Antigen provides results in just 15 minutes, making it highly suitable for quick action at the point-of-care. Anterior (shallow) nasal sampling collects from the front area of the nose and is therefore less invasive, reduces patient discomfort and simplifies the overall testing procedure.
Anterior nasal swab samples may be particularly useful for children and elderly people, especially when used for frequent testing. This simplified collection method also allows for the option for patients to self-collect under supervision, offering reduced physical contact and exposure and further protection for healthcare practitioners.
Results in 15 minutes.
-
Quick and convenient
-
Used with a shallow nasal swab
-
Locally and internationally validated

Test kit components:
-
All components are included in the kit
-
1x package insert
-
1x test cassette
-
1x sterile swab
-
1x collection tube with pre-filled test liquid
Test Procedure:
1.
Remove the test device from its packaging, placing it on a clean, flat surface.
2.
Locate the nasal swab and carefully remove it from its packaging, ensuring not to touch the swab tip.
Gently insert the swab 2 – 3cm into your nostril.
3.
Slowly rotate the swab at least 5 times in a circular path over the surface of the nostril for a total of 15 seconds.
Using the same swab, repeat on your other nostril, also for a total of 15 seconds.
4.
Locate the extraction vial and gently peel off the aluminium foil seal.
5.
Carefully place the swab into the extraction vial and rotate the swab vigorously at least 5 times.
6.
Remove the swab by rotating against the extraction vial, while squeezing the sides of the vial to release the liquid from the swab.
7.
Close the vial by firmly pushing in the cap provided.
8.
Thoroughly mix the vial by swirling and flicking the bottom of the tube.
9.
Invert the extraction vial and hold the sample vertically above the sample well on the test device.
Squeeze the vial gently and allow 3 drops of sample to fall into the sample well.
10.
Wait for 10 minutes for the results.
It is important that the test device is not moved or lifted during this time.
Results should not be read before 10 minutes or after 15 minutes.
11.
Dispose all testing components, including the test device, in an appropriate hazardous waste bin.
8.
Thoroughly mix the vial by swirling and flicking the bottom of the tube.
Reading the Results:
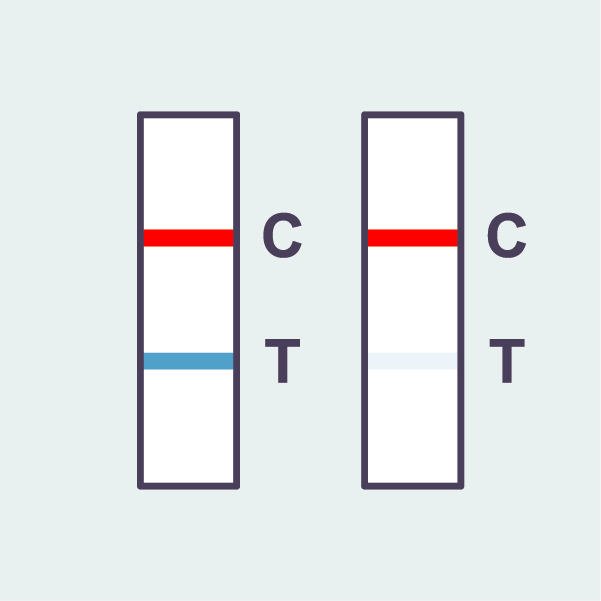
COVID-19 positive
A positive result shows 2 lines:
– A red line at ‘C’
– A blue line at ‘T’
Any faint coloured line should
be considered as positive.
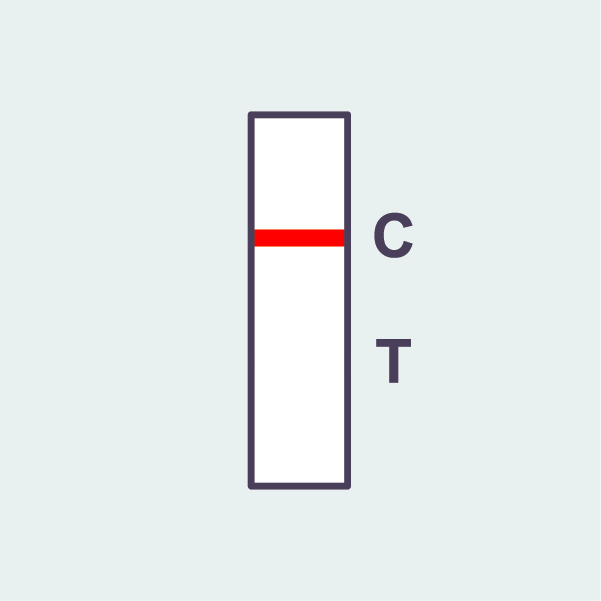
COVID-19 negative
A negative result shows:
– ONE (1) red line at ‘C’.
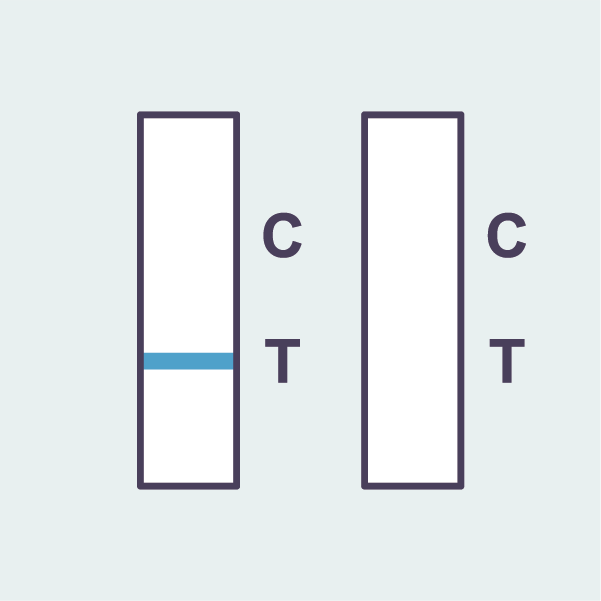
Inconclusive result
If the red line is not visible, the result is invalid. Another test will be required.
Squeeze the vial gently and allow 3 drops of sample to fall into the sample well.
In the case of a positive result, you must immediately:
1.
Undergo a laboratory-based
PCR test.
2.
Contact your local Public Health Unit and follow their instructions.
3.
Self-isolate until you receive
a negative PCR test result
or as instructed.
If you would like more information on the above products or would like to place a trade order, please fill in the contact form below.